Jj thomson cathode ray tube experiment year12/31/2023 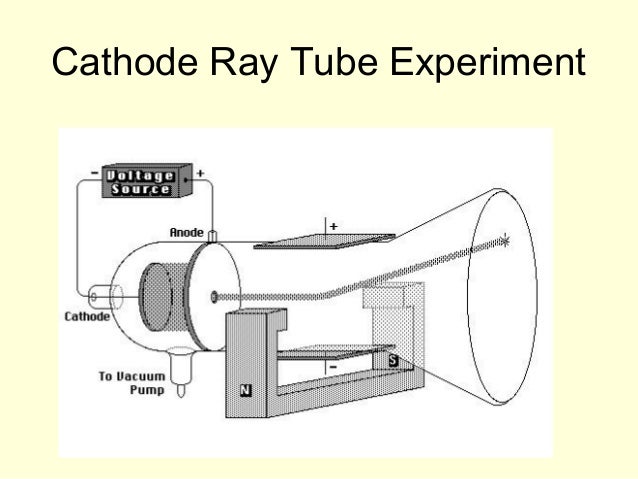 
This showed that cathode rays consist of negatively charged particles. He observed that cathode rays were deflected towards the positive plate of the electric field. Thomson applied an electric field in the path of cathode rays in the discharge tube. DISCOVERY OF ELECTRONS – THOMSON EXPERIMENT AND RESULT He discovered that cathode rays consist of negatively charged subatomic particles (now called electrons), present in all atoms of the elements. (c) In the cathode ray, the beam (shown in yellow) comes from the cathode and. (b) This is an early cathode ray tube, invented in 1897 by Ferdinand Braun. Thomson produced a visible beam in a cathode ray tube. He could then calculate the charge-to-mass ratio (e/m) of the electron. The magnetic field deflected the electrons into circular paths of known radius (r). J Thomson discovered electrons while studying characteristics of cathode rays. We shall trace his efforts that established that the cathode rays are actually a stream of particles carrying negative electrical charges. The results of these measurements indicated that these particles were much lighter than atoms (Figure 3.3. Thomson used an electric field (V) to accelerate electrons into a magnetic field (B). Although, he could not establish what electric current or cathode rays comprise and what is moving from the negative terminal to the positive terminal.įinally, in 1897 J. Thomson published this work in 1897, for which he received much acclaim - he was awarded the Nobel Prize for physics in 1906 for this work, and was knighted in 1908. William observed that when he applies a high electric potential across the discharge tube, then current flows from the negative terminal to the positive terminal in the form of rays. In a series of experiments using cathode ray tubes, Thomson concluded that cathode rays were particles with a negative charge and much smaller in size than an atom. His work involved the use of cathode ray tubes and identifying a. Thomson, in 1898, proposed that an atom possesses a. J.J Thomson contributed massively to the model of the atom and the modern day theory. William Crooke, the British Scientist, in the year 1875 successfully created a near to perfect vacuum (0.01 mm of mercury) in a glass tube sealed at both ends with metal plates. To vacuum pump cathode cathode High voltage A cathode ray discharge tube. William Crook is the first person to pass an electric current through a vacuum Each kind of ion has its own characteristic charge to mass ratio, because each ion has a specific electrical charge and its own characteristic mass. However, most of them failed as none of them were able to create a perfect vacuum. Kaufmann is worth mentioning in an account of the discovery of the electron because he used the very method described here by Thomson to measure of cathode rays in 1897. After carrying out a series of experiments with – solids, liquids, and gases, scientists took one step further to drive electricity through a vacuum. They proved that the electricity or the electric current can flow through any substance – solid, liquid or gas if enough driving force or electric potential exists. The magnitude of the electric current flowing through a substance is directly proportional to the electric potential applied across it. The electric potential is a driving force that results in the flow of current through a substance due to the difference in concentration of charges at two ends of it. Scientists in the early nineteenth century were aware of electricity and the effect of electric potential. Who discovered electrons CATHODE RAY EXPERIMENT To know about the basic structure of an atom, click here Perrin had found that cathode rays deposited an. These cylinders were in turn connected to an electrometer, a device for catching and measuring electrical charge. This led to the discovery of the nucleus and other subatomic particles – protons and neutrons. Three experiments led him to this.: irst, in a variation of an 1895 experiment by Jean Perrin, Thomson built a cathode ray tube ending in a pair of metal cylinders with a slit in them. Thomson discovered electrons, the race began among other scientists to uncover the basic structure of an atom. 
In his quest to study properties of cathode rays, he discovered that atoms contain negatively charged subatomic particles – ‘electrons’. But things changed after the discovery made by an English scientist named J.J. Thomson, the one who discovered electronsįor a long period in history, scientists were of opinion that atoms could not be broken further. In 1897, Thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an. 
As regards the radiant efficiency, that was found to be. Scientists had now established that the atom was not indivisible as Dalton had believed, and due to the work of Thomson, Millikan, and others, the charge and mass of the negative, subatomic particles-the electrons-were known.J. cathode rays and the particles discharged from negatively - charged bodies.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
